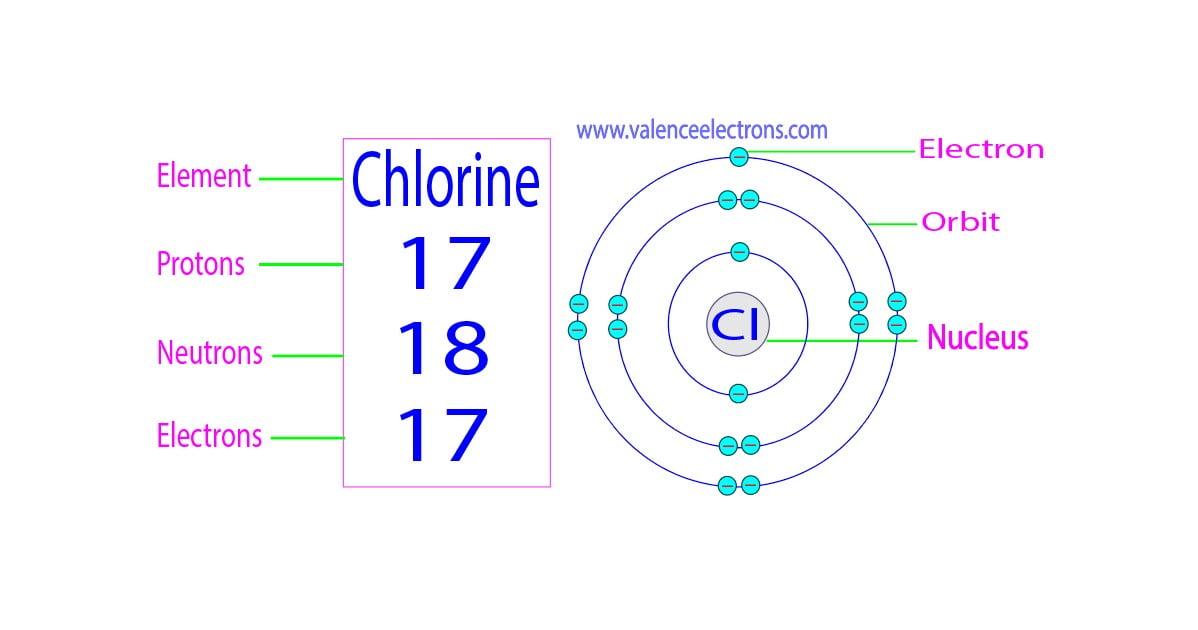
Chlorine-35 Protons . It has an atomic weight of 35.450 and a mass. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes having 15 to 23 neutrons. Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. Only two chlorine isotopes exist in significant. Only two chlorine isotopes exist in. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes with 15 to 23 neutrons. There are two stable isotopes, 35 cl. 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl.
from chamotgallery.com
Only two chlorine isotopes exist in significant. 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. It has an atomic weight of 35.450 and a mass. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes with 15 to 23 neutrons. Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. There are two stable isotopes, 35 cl. Only two chlorine isotopes exist in. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes having 15 to 23 neutrons.
How many protons, neutrons and electrons does chlorine have? (2023)
Chlorine-35 Protons 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. It has an atomic weight of 35.450 and a mass. Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. Only two chlorine isotopes exist in. 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes having 15 to 23 neutrons. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes with 15 to 23 neutrons. There are two stable isotopes, 35 cl. Only two chlorine isotopes exist in significant.
From www.alamy.com
Chlorine atomic structure Cut Out Stock Images & Pictures Alamy Chlorine-35 Protons Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. It has an atomic weight of 35.450 and a mass. All atoms of chlorine (cl) have 17. Chlorine-35 Protons.
From askfilo.com
Average atomic mass of chlorine is 35.5u. It has two isotopes of atomic m.. Chlorine-35 Protons It has an atomic weight of 35.450 and a mass. Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. There are two stable isotopes, 35 cl. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes having 15 to 23 neutrons. Only two chlorine isotopes exist. Chlorine-35 Protons.
From www.sciencephoto.com
Isotopes of chlorine, illustration Stock Image C028/6461 Science Chlorine-35 Protons 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. It has an atomic weight of 35.450 and a mass. Only two chlorine isotopes exist in significant. Only two chlorine isotopes exist in. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes having. Chlorine-35 Protons.
From www.dreamstime.com
Chlorine chemical element stock vector. Illustration of proton 83100744 Chlorine-35 Protons 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. Only two chlorine isotopes exist in significant. All atoms of chlorine (cl) have 17 protons, but there. Chlorine-35 Protons.
From www.chegg.com
Solved 14. Which statement describes chlorine35? a. 17 Chlorine-35 Protons All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes having 15 to 23 neutrons. Only two chlorine isotopes exist in. 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. It has an atomic weight of 35.450 and a mass. All atoms of. Chlorine-35 Protons.
From sciencenotes.org
Argon Facts Chlorine-35 Protons Only two chlorine isotopes exist in. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes with 15 to 23 neutrons. Only two chlorine isotopes exist in significant. Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. 53 rows chlorine (17 cl) has 25 isotopes, ranging. Chlorine-35 Protons.
From slideplayer.com
ATOMS Standard C2 Students will demonstrate an understanding of atomic Chlorine-35 Protons There are two stable isotopes, 35 cl. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes having 15 to 23 neutrons. It has an atomic weight of 35.450 and a mass. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes with 15 to 23 neutrons. Only two chlorine isotopes exist in. Chlorine. Chlorine-35 Protons.
From brokeasshome.com
Chlorine Periodic Table Protons Neutrons Electrons Chlorine-35 Protons Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes having 15 to 23 neutrons. It has an atomic weight of 35.450 and a mass. Only two chlorine isotopes exist in significant. There are two stable isotopes,. Chlorine-35 Protons.
From all-about-chlorine.weebly.com
Composition and Structure All About Chlorine Chlorine-35 Protons Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes with 15 to 23 neutrons. 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m. Chlorine-35 Protons.
From www.nuclear-power.com
Chlorine Atomic Number Atomic Mass Density of Chlorine nuclear Chlorine-35 Protons Only two chlorine isotopes exist in significant. Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. It has an atomic weight of 35.450 and a mass. 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl.. Chlorine-35 Protons.
From brainly.in
Explain why chlorine always has relative atomic mass of about 35.5u Chlorine-35 Protons 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. It has an atomic weight of 35.450 and a mass. There are two stable isotopes, 35 cl.. Chlorine-35 Protons.
From brokeasshome.com
Chlorine Periodic Table Protons Neutrons Electrons Chlorine-35 Protons Only two chlorine isotopes exist in. Only two chlorine isotopes exist in significant. 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. It has an atomic weight of 35.450 and a mass. There are two stable isotopes, 35 cl. All atoms of chlorine (cl) have 17. Chlorine-35 Protons.
From mckeethtaryn.blogspot.com
Online Chem Class 2012 Activity 2 Chlorine-35 Protons Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. Only two chlorine isotopes exist in significant. Only two chlorine isotopes exist in. All atoms of chlorine. Chlorine-35 Protons.
From greekchlist.weebly.com
Chlorine atomic mass greekchlist Chlorine-35 Protons 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. There are two stable isotopes, 35 cl. Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. Only two chlorine isotopes exist in. All atoms of chlorine. Chlorine-35 Protons.
From kennedykruwliu.blogspot.com
How Many Protons Does Chlorine 35 Have KennedykruwLiu Chlorine-35 Protons All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes with 15 to 23 neutrons. It has an atomic weight of 35.450 and a mass. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes having 15 to 23 neutrons. Only two chlorine isotopes exist in significant. There are two stable isotopes, 35 cl.. Chlorine-35 Protons.
From chamotgallery.com
How many protons, neutrons and electrons does chlorine have? (2023) Chlorine-35 Protons Only two chlorine isotopes exist in. Only two chlorine isotopes exist in significant. Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. All atoms of chlorine. Chlorine-35 Protons.
From brainly.in
draw atomic structure of chlorine Brainly.in Chlorine-35 Protons Only two chlorine isotopes exist in significant. 53 rows chlorine (17 cl) has 25 isotopes, ranging from 28 cl to 52 cl, and two isomers, 34m cl and 38m cl. Only two chlorine isotopes exist in. It has an atomic weight of 35.450 and a mass. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes with. Chlorine-35 Protons.
From www.slideserve.com
PPT How many protons, neutrons, and electrons are there in an atom of Chlorine-35 Protons Only two chlorine isotopes exist in. Chlorine is the 17th element in the periodic table and has a symbol of cl and atomic number of 17. All atoms of chlorine (cl) have 17 protons, but there are chlorine isotopes having 15 to 23 neutrons. Only two chlorine isotopes exist in significant. There are two stable isotopes, 35 cl. It has. Chlorine-35 Protons.